Brazil Angioplasty Devices Market Size, Share, and COVID-19 Impact Analysis, By Product Type (Stents, Balloons, Guidewires, and Others), By Application (Peripheral Angioplasty and Coronary Angioplasty), and Brazil Angioplasty Devices Market Insights, Industry Trend, Forecasts to 2033
Industry: HealthcareBrazil Angioplasty Devices Market Insights Forecasts to 2033
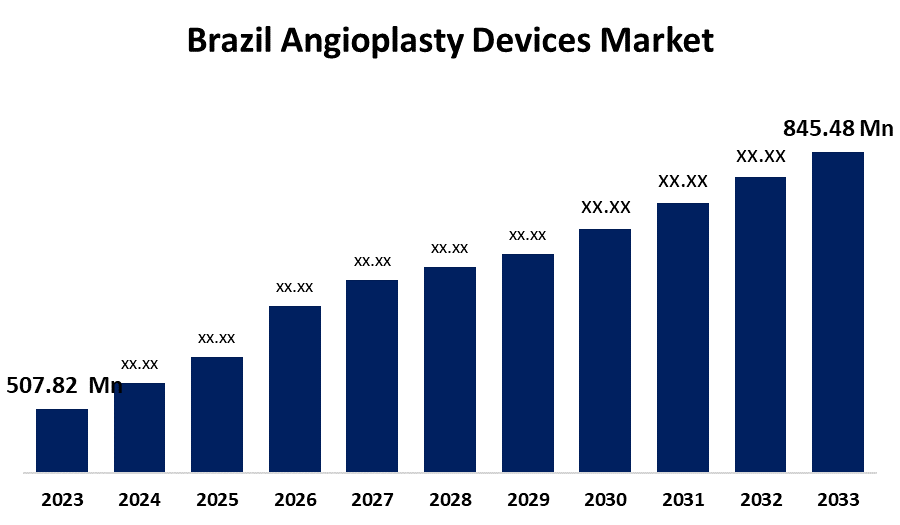
- The Brazil Angioplasty Devices Market Size Was Estimated at USD 507.82 Million in 2023.
- The Market is Growing at a CAGR of 5.23% from 2023 to 2033
- The Brazil Angioplasty Devices Market Size is Expected to Reach USD 845.48 Million by 2033
Get more details on this report -
The Brazil Angioplasty Devices Market Size is expected to reach USD 845.48 million by 2033, growing at a CAGR of 5.23% from 2023 to 2033.
Market Overview
The market for angioplasty devices in Brazil is the area of the medical device industry dedicated to instruments and apparatus used in angioplasty operations. To restore appropriate blood flow, angioplasty is a minimally invasive procedure used to open constricted or blocked blood vessels, usually arteries. This market serves both coronary and peripheral angioplasty applications and comprises goods such as stents, guidewires, balloons, and other associated equipment. In addition to addressing the rising incidence of cardiovascular disorders in Brazil, it provides services to hospitals, specialist clinics, and other healthcare facilities. The market is expanding significantly as a result of the growing number of elderly citizens, the increased prevalence of cardiovascular disease, and the growing use of minimally invasive procedures. Additionally, the healthcare system in Brazil is changing dramatically as a result of large investments and modernization projects. Furthermore, government initiatives aid the market expansion, for instance in August 2024, the Ministry of Health released new guidelines to expedite regulatory procedures and facilitate the market introduction of cutting-edge medical devices, like angioplasty instruments.
Report Coverage
This research report categorizes the market for the Brazil angioplasty devices market based on various segments and regions and forecasts revenue growth and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing Brazil angioplasty devices market. Recent market developments and competitive strategies such as expansion, product launch, and development, partnership, merger, and acquisition have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the Brazil angioplasty devices market.
Brazil Angioplasty Devices Market Report Coverage
| Report Coverage | Details |
|---|---|
| Base Year: | 2023 |
| Market Size in 2023 : | USD 507.82 million |
| Forecast Period: | 2023 - 2033 |
| Forecast Period CAGR 2023 - 2033 : | 5.23% |
| 2033 Value Projection: | USD 845.48 million |
| Historical Data for: | 2019-2022 |
| No. of Pages: | 220 |
| Tables, Charts & Figures: | 109 |
| Segments covered: | By Application, By Product Type |
| Companies covered:: | Abbott Laboratories, Boston Scientific Corporation, B.Braun SE, Becton, Dickinson, and Company, MicroPort Scientific Corporation, SCITECH, Terumo Corporation, Biotronik SE & Co. KG, Medtronic PLC, Others, |
| Pitfalls & Challenges: | Covid-19 Empact, Challenges, Growth, Analysis. |
Get more details on this report -
Driving Factors
The angioplasty devices market in Brazil is expanding due to increasing cardiovascular diseases and medical advancements. As people age and lifestyle changes, the demand for treatment options for coronary artery disease increases. Additionally, investing in research and development is essential to advancing angioplasty technology in Brazil, where producers are creating cutting-edge tools that enhance patient outcomes and procedure efficiency. In addition, the market is expanding further because of technological developments such as drug-eluting stents and bioresorbable scaffolds, which improve patient outcomes and satisfy the need for sophisticated cardiovascular treatments.
Restraining Factors
The market for angioplasty devices in Brazil is hampered by issues like the high price of sophisticated devices, the lack of access to healthcare in rural areas, and the scarcity of qualified personnel for intricate procedures. Furthermore, regulatory obstacles and unstable economies might postpone product approvals, which can affect patient accessibility and market expansion.
Market Segmentation
The Brazil angioplasty devices market share is classified into product type and application.
- The stents segment accounted for the largest share in 2023 and is expected to grow at a significant CAGR during the forecast period.
Based on the product type, the Brazil angioplasty devices market is divided into stents, balloons, guidewires, and others. Among these, the stents segment accounted for the largest share in 2023 and is expected to grow at a significant CAGR during the forecast period. The country's increased use of these devices in angioplasty treatments is the reason for this noteworthy market position. In addition to technological developments in stent designs and materials, the segment's leadership is further supported by the growing number of coronary artery disorders that require stent implantation.
- The coronary angioplasty segment accounted for the highest share in 2023 and is expected to grow at a significant CAGR during the projected timeframe.
Based on the application, the Brazil angioplasty devices market is classified into peripheral angioplasty and coronary angioplasty. Among these, the coronary angioplasty segment accounted for the highest share in 2023 and is expected to grow at a significant CAGR during the projected timeframe. The high prevalence of coronary artery disease in Brazil and the growing popularity of minimally invasive procedures are the primary factors driving this noteworthy market position. In addition, technological developments in coronary angioplasty treatments, such as the use of artificial intelligence to improve stent placement precision, are another factor contributing to the segment's rise.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the Brazil angioplasty devices market along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, business strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Abbott Laboratories
- Boston Scientific Corporation
- B.Braun SE
- Becton, Dickinson, and Company
- MicroPort Scientific Corporation
- SCITECH
- Terumo Corporation
- Biotronik SE & Co. KG
- Medtronic PLC
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting And Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In September 2023, for the first time, the ReewarmTM PTX Drug-Coated Balloon Catheter was successfully implanted in Brazil. The femoral-popliteal artery stenosis can be treated with this device by minimally invasive treatments.
Market Segment
This study forecasts revenue at Brazil, regional, and country levels from 2020 to 2033. Spherical Insights has segmented the Brazil angioplasty devices market based on the below-mentioned segments:
Brazil Angioplasty Devices Market, By Product Type
- Stents
- Balloons
- Guidewires
- Others
Brazil Angioplasty Devices Market, By Application
- Peripheral Angioplasty
- Coronary Angioplasty
Need help to buy this report?